ThinPrep® Pap Test
1st
FDA-approved collection media for use with all FDA-approved HPV tests.*
A Wealth of Knowledge in a Single Vial.
Confidence in Every Step of the Process
Comprehensive Out-of-the Vial Testing
FDA Approved and Cleared Out-of-the Vial Testing
- Aptima® HPV Assay
- Aptima® HPV 16 18/45 Genotype Assay
- Alinity HPV Test
- Cobas® HPV Test
- BD Onclarity HPV Test
- Hybrid Capture® 2 HPV Test
- CINtec PLUS Cytology
- Aptima Combo 2® Assay
- BD CT/NG Assay
- Cobas® CT/NG Assay
- Aptima® Trichomonas vaginalis Assay§
- Roche TV/MG§
Comparing Sample Integrity and Preservation


Versatile Application, Increased Detection

|
SurePath™ Pap Test11 |
|
|---|---|---|
| Detection | Significantly more effective than conventional Pap smear for the detection of LSIL and more severe lesions | Similar to conventional pap smear |
| FDA-approved labelling for improved ability to detect glandular disease | ||
| Preservative Solution | PreservCyt® Solytion, Methanol based | SurePath® Solution, Ethanol / Formaldehyde based |
| Pro-Cytology volume available for OTV | 4 mL pre-cytology | 0.5 mL pre-cytology |
| Formaldehyde Free | Contains Formaldehyde DNA and RNA cross-linking2,12 | |
| Adjunctive Trichomonas vaginalis Clearance5 | ||
| Cytology Stability | 6 weeks for cytology | 4 weeks for cytology13 |
| Most adjunctive FDA approvals / clearances from a single vial |
Compared Disease Detection between ThinPrep and SurePath Solutions: Real World US Data
Two U.S. academic studies published in 2019 and 2021 compared disease detection rates between TP and SP:
- ThinPrep demonstrated a higher HSIL detection rate than SurePath14
- ThinPrep detected considerably more AGCs than SurePath15
A Thorough Approach to Cervical Cancer Detection16
While squamous cell carcinoma has steadily decreased since the introduction of the Pap smear, adenocarcinoma remains an area of great concern. The ability to detect adenocarcinoma is an essential part of comprehensive cervical cancer screening.
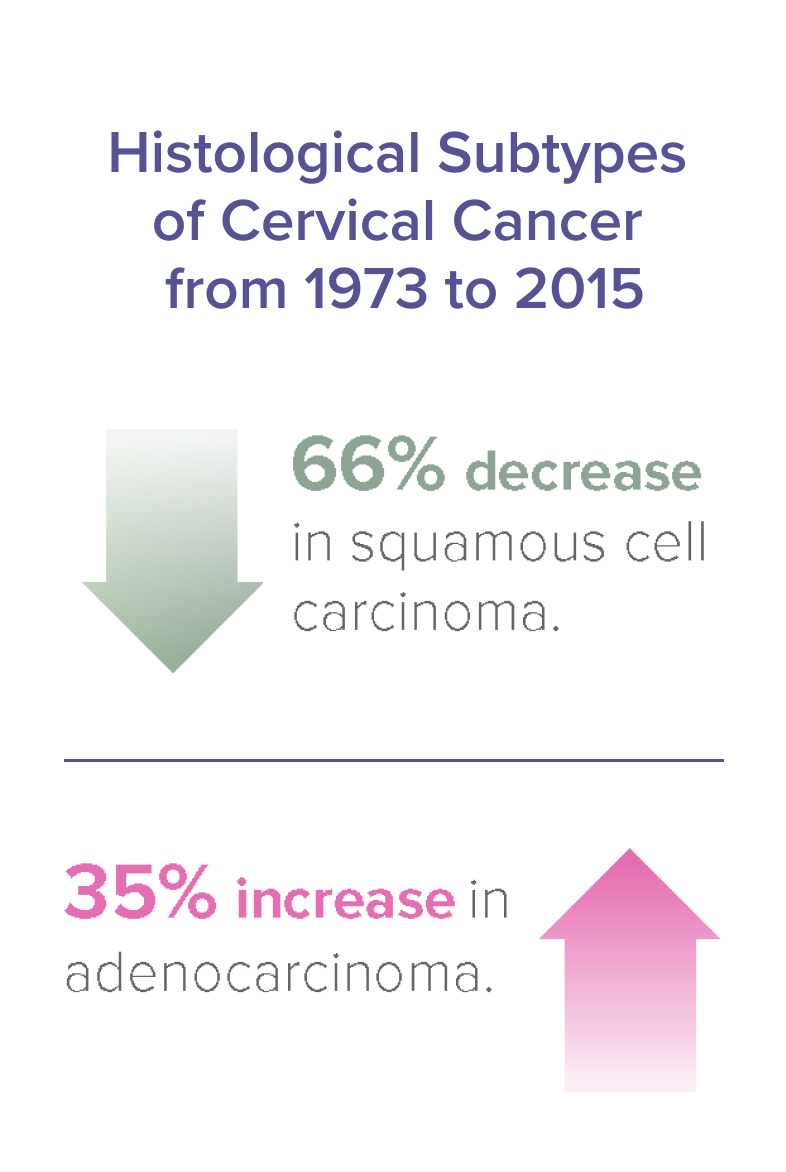
ThinPrep Improves Sensitivity by 56.76% vs. Conventional Pap Test17
Imaging Raises the Bar in Pap Testing Results
Studies have reported reductions in unsatisfactory results when the ThinPrep® imaging system was implemented.18,19
A Step Ahead: Utilizing the ThinPrep Imaging System in Combination with the ThinPrep Pap Test
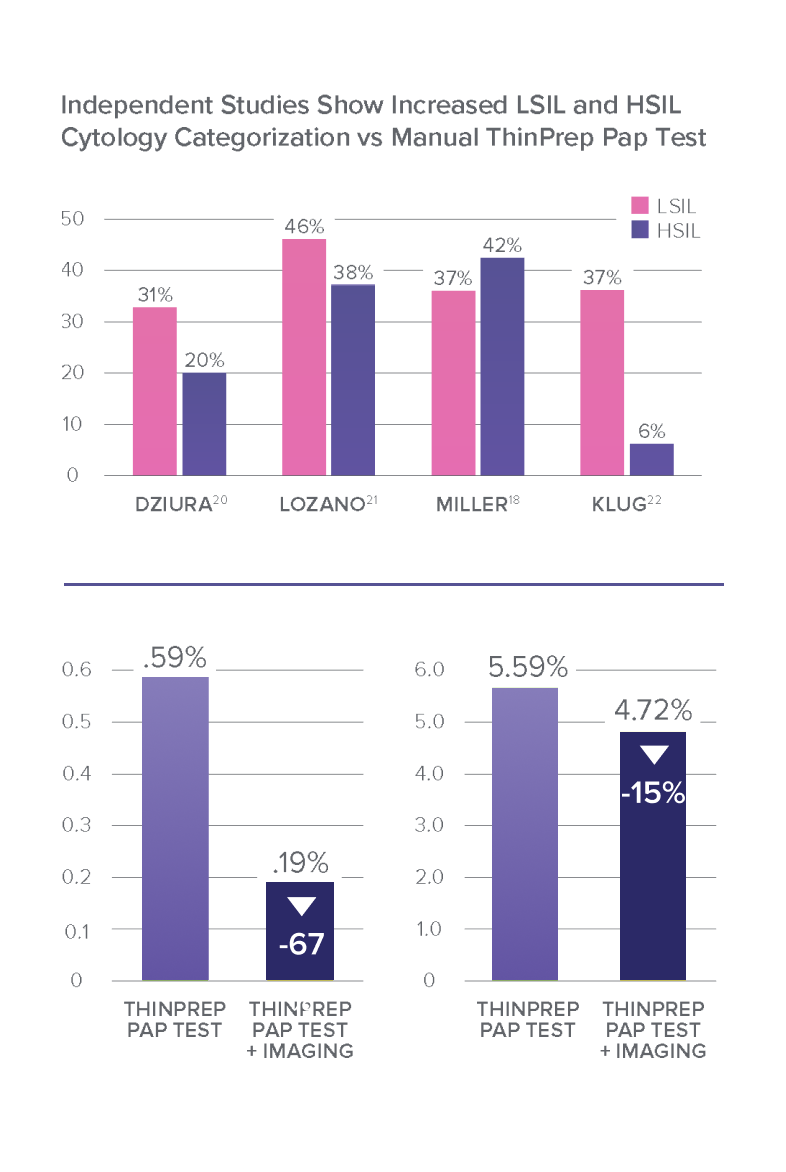
Imaging-Directed Cytology Improves Patient Results23
Increased sensitivity and specificity over manually reviewed ThinPrep Pap test slides.8
Improved standardization at each stage of sample processing.
Reduce false-negative fraction
Targeted areas: Imager identifies largest and darkest nuclei for review.
Let’s Connect
Have a question or need to talk to a Hologic team member? We’re here to help.
