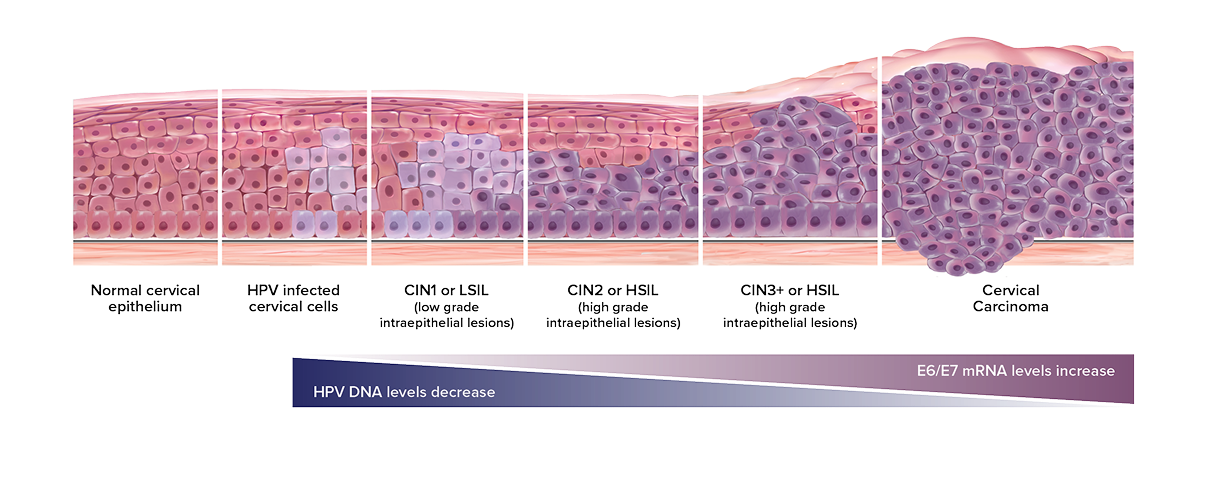
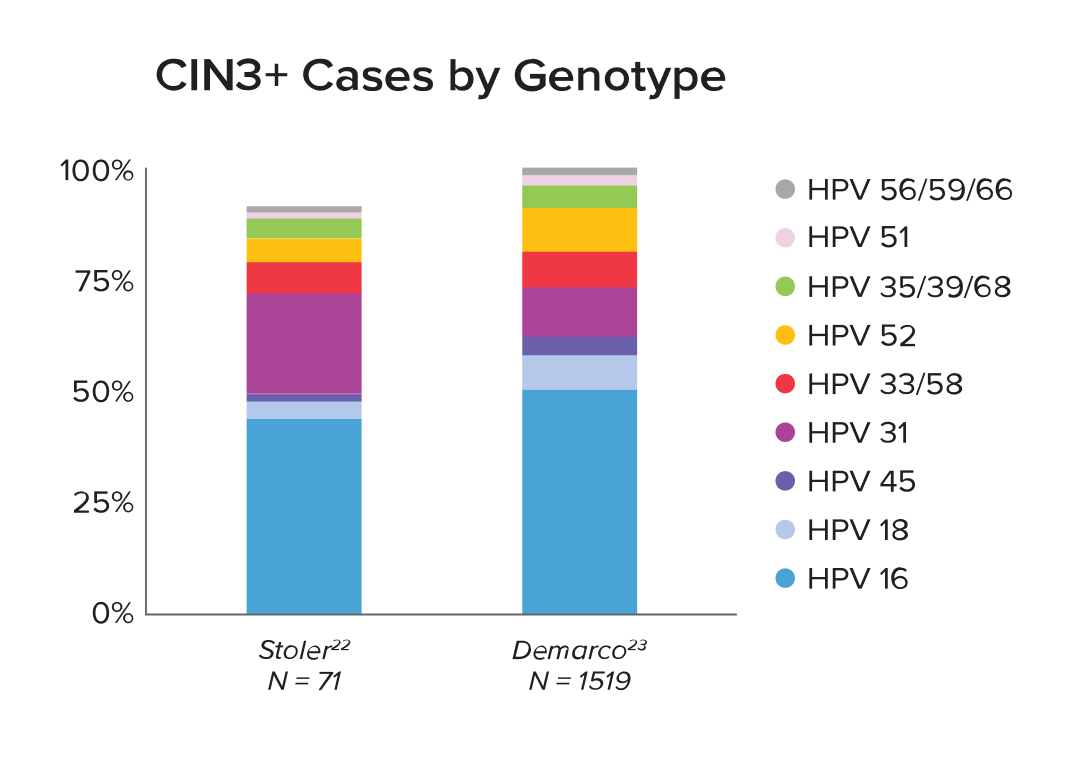
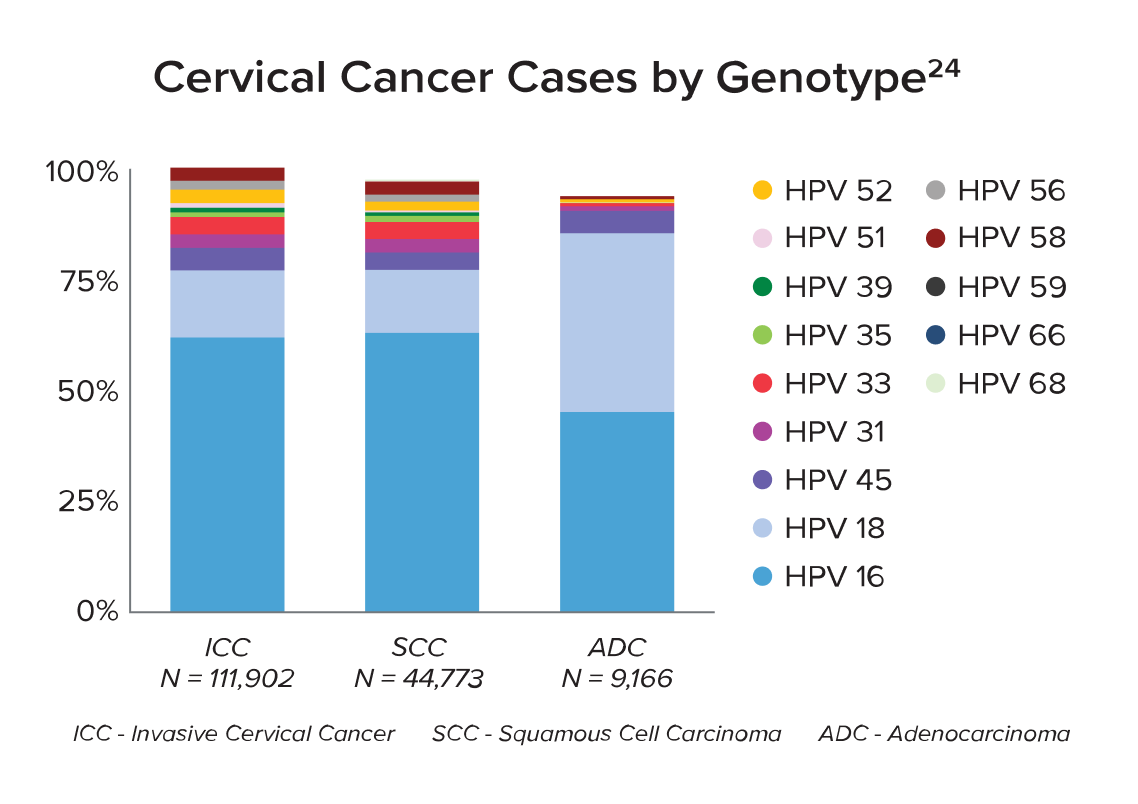
* As
described
in the
2019
ASCCP
guidelines,
collection
of
cervical
cytology
at the
colposcopy
visit is
recommended
because
additional
diagnostic
testing
and
surveillance
are
required.
†
In
certain
circumstances,
patients
who test
positive
for HPV
59, 56,
or 66 may
be
referred
to
one-year
follow-up
without
cytology/dual
stain
triage.
1.Tinelli
A, et al.
HPV viral
activity
by mRNA
HPV
molecular
analysis
to screen
the
transforming
infections
in
precancer
cervical
lesions.
Curr
Pharm
Biotechnol.
2009;10(8):767-771.
2.
Cuschieri
K, et al.
Human
Papillomavirus
Type
Specific
DNA and
RNA
Persistence–Implications
for
Cervical
Disease
Progression
and
Monitoring.
J Med
Virol.
2004;73(1):65-70.doi:10.1002/jmv.20062.
3.
Aptima
HPV Assay
[package
insert].
AW-12820,
Rev 004.
San
Diego,
CA:
Hologic,
Inc.;
2020.
4.
Rebolj M,
et al.
Extension
of
cervical
screening
intervals
with
primary
human
papillomavirus
testing:
observational
study of
English
screening
pilot
data BMJ
2022; 377
:e068776
doi:10.1136/bmj-2021-068776.
5.
Doorbar
J.
Molecular
biology
of human
papillomavirus
infection
and
cervical
cancer.
Clin Sci
(Lond).
2006
May;110(5):525-41.
doi:
10.1042/
CS20050369.
PMID:
16597322.
6.
Rebolj et
al. A
daunting
challenge:
Human
Papillomavirus
assays
and
cytology
in
primary
cervical
screening
of women
below age
30 years.
EU J of
Cancer
(2015)
51,
1456-1466.
7.
Saslow D,
et al.
American
Cancer
Society,
American
Society
for
Colposcopy
and
Cervical
Pathology,
and
American
Society
for
Clinical
Pathology
Screening
Guidelines
for the
Prevention
and Early
Detection
of
Cervical
Cancer.Am
J Clin
Pathol
2012;137:516-542.
8. Wu
R, et al.
Human
papillomavirus
messenger
RNA assay
for
cervical
cancer
screening:
the
Shenzhen
Cervical
Cancer
Screening
Trial I.
Int J
Gynecol
Cancer.
2010;20(8):1411-1414.
9.
Ratnum S,
et al.
Aptima
HPV E6/E7
mRNA test
is as
sensitive
as hc2
Assay but
more
specific
at
detecting
cervical
precancer
and
cancer. J
Clin
Microbiol.
2011;49(2):557-564.
10.
Monsonego
J, et al.
Evaluation
of
oncogenic
human
papillomavirus
RNA and
DNA tests
with
liquid-based
cytology
in
primary
cervical
cancer
screening:
the FASE
study.
Int J
Cancer.
2011,129(3):691-701.
11.
Iftner T,
et al.
GAST:
German
Aptima
Screening
Trial.
Comparison
of Aptima
and hc2
in
routine
screening
in
Germany.
Symposium
presentation
at
EUROGIN
2012. 12.
Cuzick J,
et al.
Comparing
the
performance
of six
human
papillomavirus
tests in
a
screening
population.
British J
Cancer.
2013;108:908-913.
12.
Nieves L,
et al.
Primary
Cervical
Cancer
Screening
and
Triage
Using an
mRNA
Human
Papillomavirus
Assay and
Visual
Inspection.
Int J
Gynecol
Cancer.
2013;23:513-518.
13.
Iftner T,
et al.
Head-to-Head
Comparison
of the
RNA-Based
Aptima
Human
Papillomavirus
(HPV)
Assay and
the
DNA-Based
Hybrid
Capture 2
HPV Test
in a
Routine
Screening
Population
of Women
Aged 30
to 60
Years in
Germany.
J Clin
Microbiol.
2015;53(8):2509-2516.
14.
Muangto
T, et al.
Experience
of
combined
liquid
based
cervical
cytology
and
high-risk
HPV mRNA
for
cervical
cancer
screening
in
Thammasat
University
Hospital.
Asian Pac
J Cancer
Prev.
2016;17(9):4409-4413.
15.
Reid et
al. Human
Papillomavirus
Oncogenic
mRNA
Testing
for
Cervical
Cancer
Screening.
Am J Clin
Pathol,
2015;144:473-483.
16.
Cook et
al.,
Aptima
HPV Assay
versus
Hybrid
Capture®
2 HPV
test for
primary
cervical
cancer
screening
in the
HPV FOCAL
trial J.
Clin.
Virol.
2017;87:23–29.
17.
Cook et
al. Cobas
4800 HPV
and
Hybrid
Capture 2
comparison
at
baseline
and 48
months in
the HPV
Focal
trial.
Poster
presented
at IPV
2017.
18.
White C,
et al.
Performance
of the
HPV E6/E7
mRNA
Aptima
HPV assay
combined
with
partial
genotyping
compared
with the
HPV DNA
Cobas
4800 HPV
test for
use in
primary
screening:
Results
from the
CERVIVA
HPV
primary
screening
study in
Ireland
[published
online
ahead of
print,
2023 Aug
26]. Int
J Cancer.
2023;10.1002/ijc.34685.
doi:10.1002/
ijc.34685.
19.
Pi R, Li
T, Zhang
H, Zhou
H, Yang
Y, Dai Y,
Wu Z,
Jiang M,
Chen W,
Zhu L.
The
Distribution
of HR-HPV
E6/E7 DNA
and mRNA
by
Histological
Grade and
the
Clinical
Performance
for
Detection
of
Cervical
Cancer
and
Precancer.
J Med
Virol.
2024
Nov;96(11):e70026.
doi:
10.1002/jmv.70026.
PMID:
39540331.
20.
Adapted
from:
Schiffman
M et al.
Human
papillomavirus
testing
in the
prevention
of
cervical
cancer. J
Natl
Cancer
Inst.
2011;103(5):368-83.
doi:10.1093/jnci/djq562.
21.
McCredie
MR, et
al.
Natural
history
of
cervical
neoplasia
and risk
of
invasive
cancer in
women
with
cervical
intraepithelial
neoplasia
3: a
retrospective
cohort
study.
Lancet
Oncol.
2008;9(5):425-34.
doi:10.1016/S1470-2045(08)70103-7.
22.
Stoler,
et al.
Stratified
risk of
high-grade
cervical
disease
using
onclarity
HPV
extended
genotyping
in women,
≥25
years of
age, with
NILM
cytology.
2019;153(1):26-33.
doi.org/10.1016/j.ygyno.2018.12.024\
23.
Demarco,
et al. A
study of
type-specific
HPV
natural
history
and
implications
for
contemporary
cervical
cancer
screening
programs.
EClinicalMed¬icine.
2020;22.
doi:
10.1016/j.eclinm.2020.100293.
24.
Wei F,
Georges
D, Man I,
Baussano
I,
Clifford
GM.
Causal
attribution
of human
papillomavirus
genotypes
to
invasive
cervical
cancer
worldwide:
a
systematic
analysis
of the
global
literature.
Lancet.
2024 Aug
3;404(10451):435-444.
doi:
10.1016/S0140-6736(24)01097-3.
PMID:
39097395
25.
Hopenhayn
C, et al.
Prevalence
of human
papillomavirus
types in
invasive
cervical
cancers
from 7 US
cancer
registries
before
vaccine
introduction.
J Low
Genit
Tract
Dis.
2014;18(2):182-189.
doi:
10.1097/LGT.0b013e3182a577c7
26.
Massad
LS,
Clarke
MA,
Perkins
RB,
Garcia F,
Chelmow
D, Cheung
LC,
Darragh
TM,
Egemen D,
Lorey TS,
Nayar R,
Newman M,
Risley C,
Smith RA,
Wentzensen
N;
Enduring
Consensus
Cervical
Cancer
Screening
and
Management
Guidelines
Committee.
Applying
Results
of
Extended
Genotyping
to
Management
of
Positive
Cervicovaginal
Human
Papillomavirus
Test
Results:
Enduring
Guidelines.
J Low
Genit
Tract
Dis. 2025
Jan 10.
doi:
10.1097/LGT.0000000000000865.
Epub
ahead of
print.
PMID:
39791481
27.
Monsonego,
et al.
Prevalence
of
high-risk
human
papilloma
virus
genotypes
and
associated
risk of
cervical
precancerous
lesions
in a
large
U.S.
screening
population:
Data from
the
ATHENA
trial.
Gynecologic
Oncology.
2015;137(1):47-54.
doi.org/10.1016/j.ygyno.2015.01.551.
28.
Perkins
RB, Guido
RS,
Castle
PE,
Chelmow
D,
Einstein
MH,
Garcia F,
Huh WK,
Kim JJ,
Moscicki
AB, Nayar
R,
Saraiya
M, Sawaya
GF,
Wentzensen
N,
Schiffman
M; 2019
ASCCP
Risk-Based
Management
Consensus
Guidelines
Committee.
2019
ASCCP
Risk-Based
Management
Consensus
Guidelines
for
Abnormal
Cervical
Cancer
Screening
Tests and
Cancer
Precursors.
J Low
Genit
Tract
Dis. 2020
Apr;24(2):102-131.
doi:
10.1097/LGT.0000000000000525.
Erratum
in: J Low
Genit
Tract
Dis. 2020
Oct;24(4):427.
doi:
10.1097/LGT.0000000000000563.
PMID:
32243307;
PMCID:
PMC7147428.