Aptima® Trichomonas vaginalis Assay (ATV)
Curable—But Still All Too Common
Trichomoniasis (“trich”) is as prevalent as chlamydia (CT) and gonorrhea (NG) combined.1 Trichomonas vaginalis (TV) infection increases risk of other STIs & HIV, and if left untreated it can lead to pelvic inflammatory disease (PID) and adverse pregnancy outcomes.2,3
26M
new sexually transmitted infections in 20181
6.9M
infections were trich1
~70%
of people do not have any signs or symptoms2
Overlapping Symptoms Can Make Diagnosis Difficult
Many different conditions may cause symptoms similar to trich, and co-infections can be common, making accurate detection of Trichomonas vaginalis imperative for effective trich treatment.4-9

Guidelines Agree on NAAT

Nucleic acid amplification testing (NAAT) is recommended for the diagnosis of Trichomoniasis.10

NAATs are highly sensitive, detecting more TV infections than wet-mount microscopy.2
- Nucleic acid amplification testing (NAAT) is recommended for the diagnosis of TV.
- Patients should be retested ~3 months after treatment because of high recurrence rates.
- For persistent infections in women, resistance testing should be considered.
- Current partners should be referred for presumptive treatment to avoid reinfection.
- All women seeking care for vaginal discharge or who have other symptoms of trichomoniasis should be tested for TV.
- Annual TV screening for all women with HIV infection is recommended.
- People receiving care in high-prevalence settings (e.g., STD clinics and correctional facilities) should consider screening.
- Asymptomatic women at increased risk for infections should consider screening.
- People with TV should be tested for other STIs, including HIV, syphilis, chlamydia and gonorrhea.
Don’t Trust a Negative Wet Mount
Wet mounts fail to detect up to 56% of TV infections.2 It can be unreliable due to the following reasons:11,12
Sensitivity and Timing:
- Relies on detecting motile forms, leading to potential false negatives.
- Delayed testing reduces accuracy as motile trichomonads decreases over time.
Interpretation Challenges:
- Variability in clinician skills affects accuracy.
- Non-motile forms may be confused with vaginal cells.
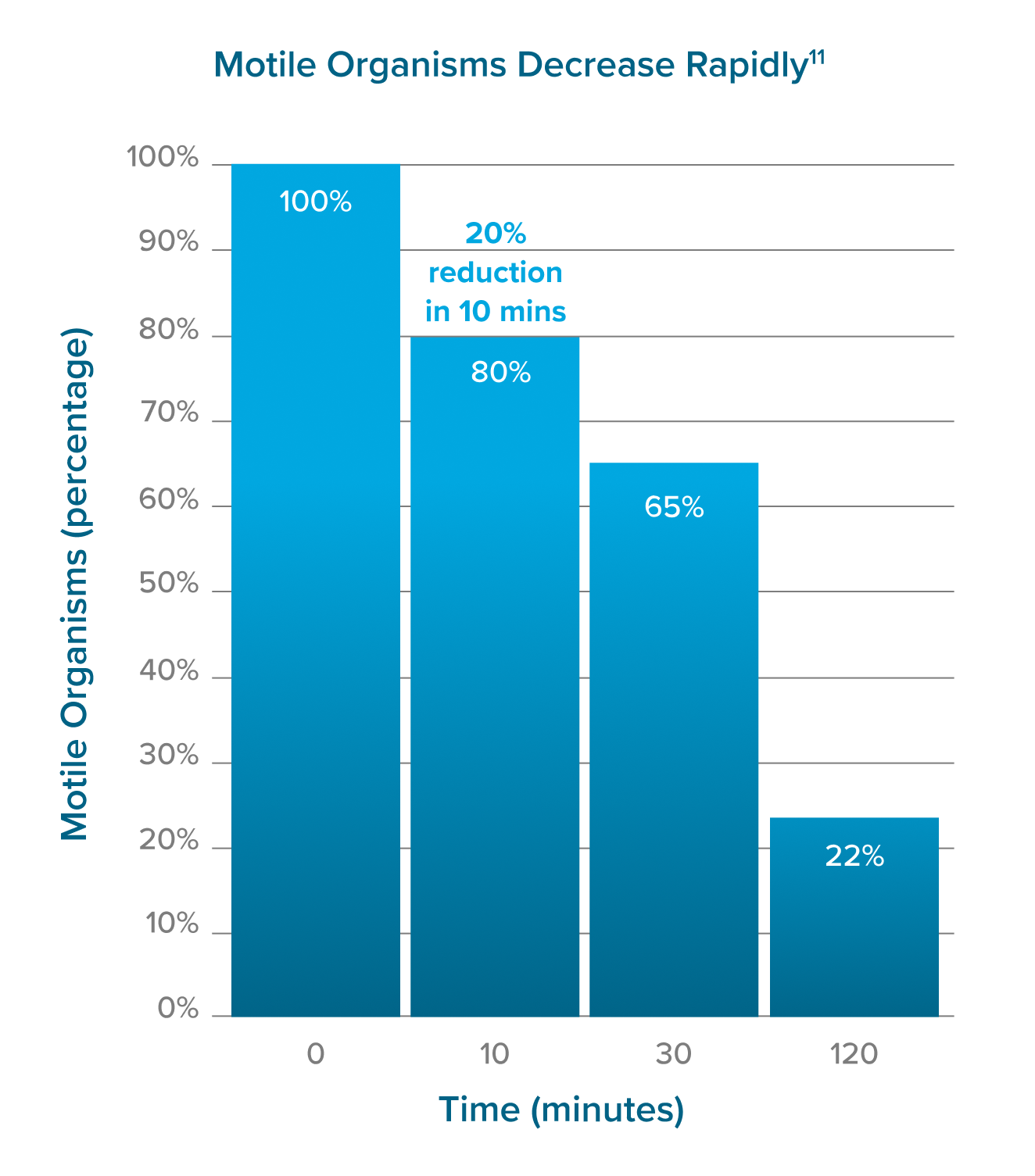
Wet mount requires at least 10,000 motile organisms/mL to visualize.12
A More Sensitive and Specific Test:
NAAT Only Requires a Fraction of One Organism to Detect up to 100% of TV Infections13
Our Aptima® Trichomonas vaginalis Assay is an FDA-cleared nucleic acid amplified test (NAAT) for both symptomatic and asymptomatic trichomoniasis case detection.
| Specimen Type | Aptima® Trichomonas vaginalis Assay (ATV)13 | ||
|---|---|---|---|
| Sensitivity | Specificity | ||

|
ThinPrep® Test Vial |
100% | 98.6% |

|
Aptima® Multitest Swab Clinician Collected Vaginal Sample |
100% | 98.2% |
|
Aptima® Multitest Swab Patient Collected Multitest Swab |
PPA 98.8% | NPA 99.4% | |
|
Aptima® Unisex Swab Endocervical Sample |
100% | 98.1% |

|
Aptima® Urine Female Sample |
100% | 100% |
|
Aptima® Urine Male Sample |
100% | 99.8% | |
Specimen Type
Aptima® Trichomonas vaginalis
Assay (ATV)13
ThinPrep® Test Vial
| Sensitivity | Specificity |
|---|---|
| 100% | 98.6% |
Aptima® Multitest Swab - Vaginal
| Sensitivity | Specificity |
|---|---|
| 100% | 98.2% |
Aptima® Multitest Swab - Patient Collected
| Sensitivity | Specificity |
|---|---|
| PPA 98.8% |
NPA 99.4% |
Aptima® Unisex Swab - Endocervical
| Sensitivity | Specificity |
|---|---|
| 100% | 98.1% |
Aptima® Urine - Female
| Sensitivity | Specificity |
|---|---|
| 100% | 100% |
Aptima® Urine - Male
| Sensitivity | Specificity |
|---|---|
| 100% | 99.8% |
Let’s Connect
Have a question or need to talk to a Hologic team member? We’re here to help.