Aptima® Combo 2 (CT/NG) Assay
A universal screening approach that protects fertility
>84% of chlamydia and gonorrhea cases are asymptomatic.1 As a result, the majority of patients are unaware that they are infected, running the risk of serious long-term health complications. Even those who are aware of their risk may not admit to being sexually active.2 Lack of screening means that roughly 30% of untreated CT infections progress to pelvic inflammatory disease (PID), while 24,000 women each year become infertile, all due to undiagnosed STIs.3,4
Improving patient care through established screening guidelines
- National health organizations have implemented screening guidelines to address increases in CT and NG prevalence.
- Nevertheless, screening implementation still faces multiple barriers such as lack of access, lack of awareness, and confidentiality concerns.
- As a result, only 59% of sexually active women ages 16–20 are screened for chlamydia per guidelines.5

CDC
Centers for Disease Control and Prevention
All sexually active women younger than 25 years should be tested for chlamydia every year.
ACOG
American College of Obstetrics and Gynecology
Screen women 24 years and younger, and older women at
increased risk.

AAP
American Academy of Pediatrics
Annual chlamydia screening of all sexually experienced females younger than 25 years.

USPSTF
U.S. Preventive Services Task Force
Screen for chlamydia in all sexually active women aged 24 years and younger and in women aged 25 years and older who are at increased risk.8

AAFP
American Academy of Family Physicians
Screen women 24 years and younger, and older women at increased risk.
The CDC now recommends that providers might consider opt-out screening for adolescent and young adult females to increase screening, be cost-saving, and identify infections among patients who do not disclose sexual behavior.
A testing solution for every young woman7
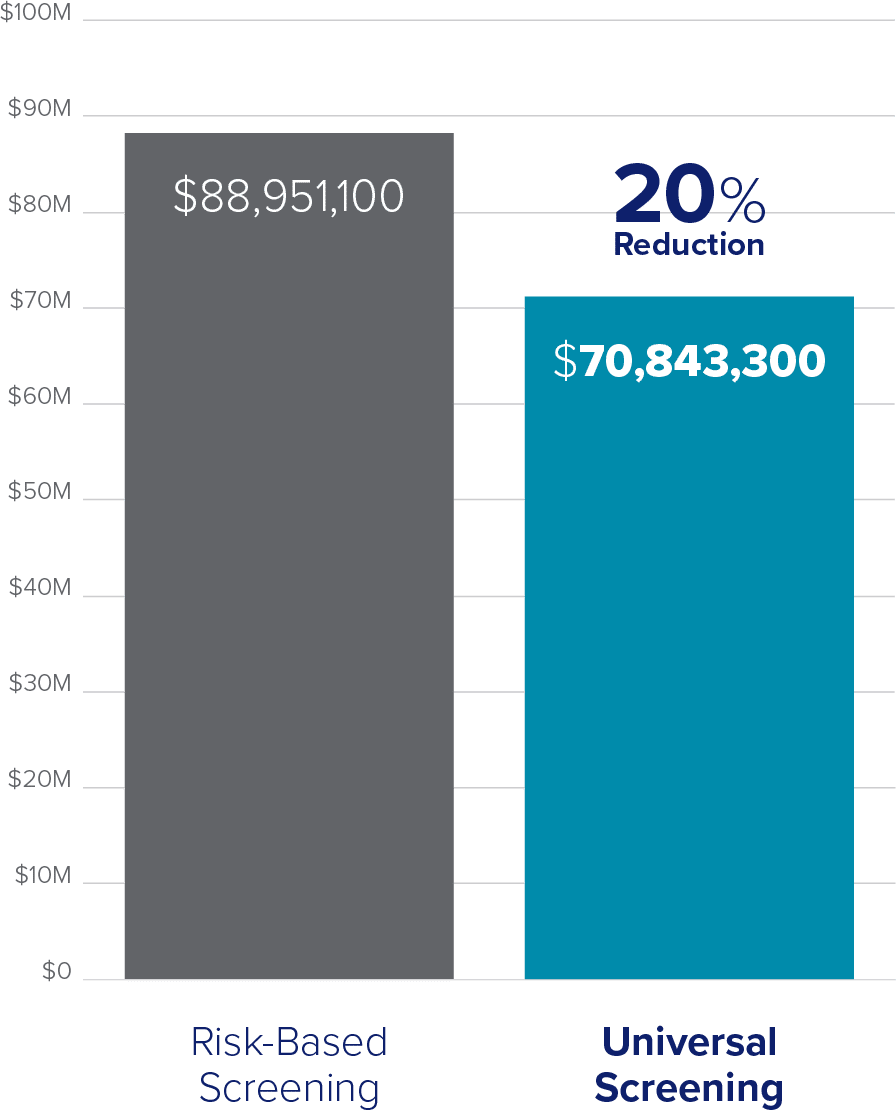
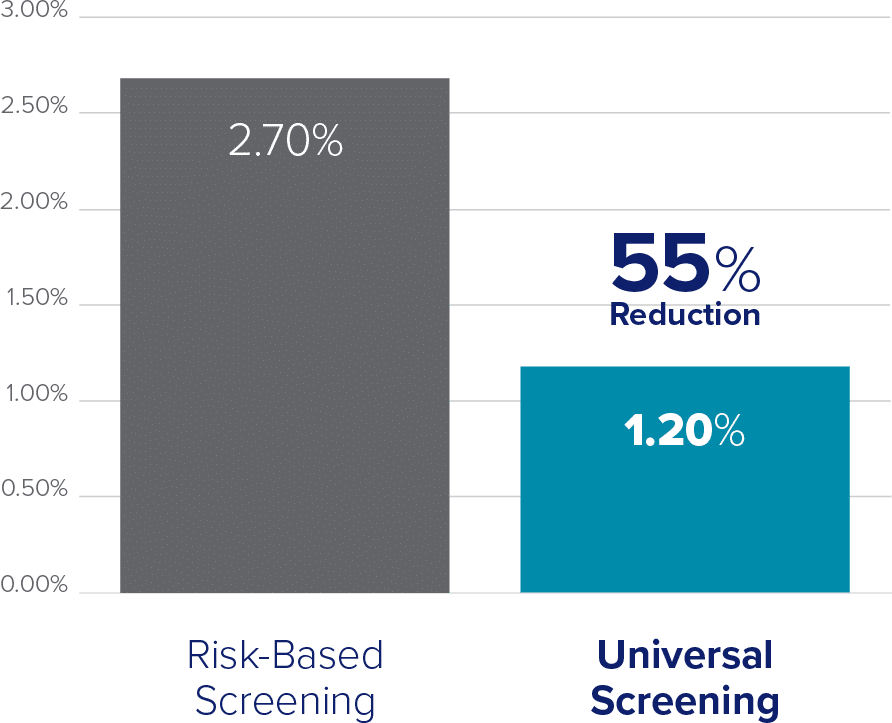
Introducing a Universal Screening protocol not only helps to prevent risk of infertility due to undiagnosed infections, but also results in decreased STI prevalence and reduces total cost to patients and healthcare organizations.
Total cost for hypothetical population of 100,000 individuals (15–24 years)
Chlamydia Prevalence
Flexible testing for accessible solutions
Introducing this new protocol is easy, starting with the way you collect a sample. Specimens collected with self and clinician-collected vaginal swabs are preferred by the CDC for CT and NG testing.9 It’s important to note that a pelvic exam is not required.
The Aptima® Multitest Swab collects one sample and provides multiple results, maximizing efficiency. Detect up to 7 infection and disease states with just one vaginal swab sample.
Testing may also be performed with any of the following alternative specimen types:
- Urine sample
- Female endocervical swab
- Liquid-based cytology specimens
- Male urethal swab
- Rectal and oropharyngeal swab
